Source: Marko Geber / Getty
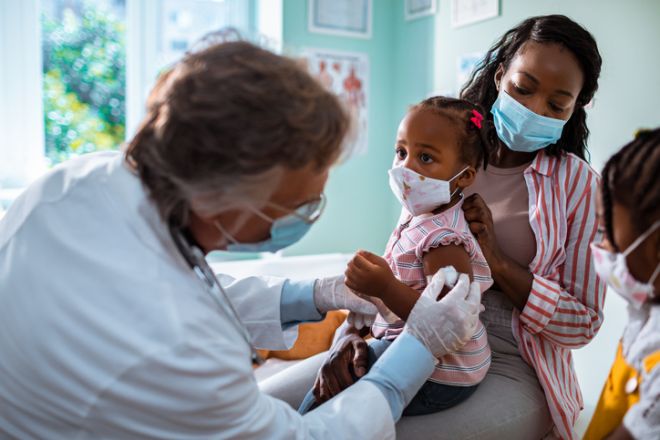
Receiving three pediatric doses of the Pfizer/BioNTech COVID-19 vaccine is 80.3 percent effective at preventing symptomatic infections and producing a strong immune response in children ages 6 months to 4 years old, according to the companies on May 23.
The new information means that in just a few weeks COVID vaccines may be authorized for the young population, after a long wait by many parents.
The data hasn’t been peer-reviewed or publicly published yet. Still, various sources report Pfizer/BioNTech pulled its research from a trial including 1,678 children amid the pandemic’s surge in Omicron cases last December.
Notably, each children’s dose is one-tenth of an adult’s dose.
Moderna’s pediatric vaccine is already being reviewed by federal officials, according to The Washington Post. The source outlined that Moderna is proposing a “two-shot regimen” of its vaccine to young children — with a 51 percent efficacy in preventing illness in those 6 months to 2 years old and 37 percent efficacy in kids 2-5 years old.
RELATED CONTENT: “Big Bird Gets Vaccinated To Encourage Children: ‘My Wing Is Feeling A Little Sore'”
The updates come after the FDA cleared the administration of the Pfizer/BioNTech vaccine booster to children ages 5-11 on May 17.
“While it has largely been the case that COVID-19 tends to be less severe in children than adults, the Omicron wave has seen more kids getting sick with the disease and being hospitalized, and children may also experience longer term effects, even following initially mild disease,” said FDA Commissioner Robert M. Califf, M.D. in a statement.
“Vaccination continues to be the most effective way to prevent COVID-19 and its severe consequences, and it is safe. If your child is eligible for the Pfizer-BioNTech COVID-19 Vaccine and has not yet received their primary series, getting them vaccinated can help protect them from the potentially severe consequences that can occur, such as hospitalization and death.”
As of now, the FDA’s Vaccines and Related Biological Products Advisory Committee plans to discuss the future and roll out of children’s COVID vaccines on June 15, NPR reports.
RELATED CONTENT: “BECAUSE YOU SHOULD KNOW: A Third Round Of Free At-Home COVID-19 Tests Are Available”









